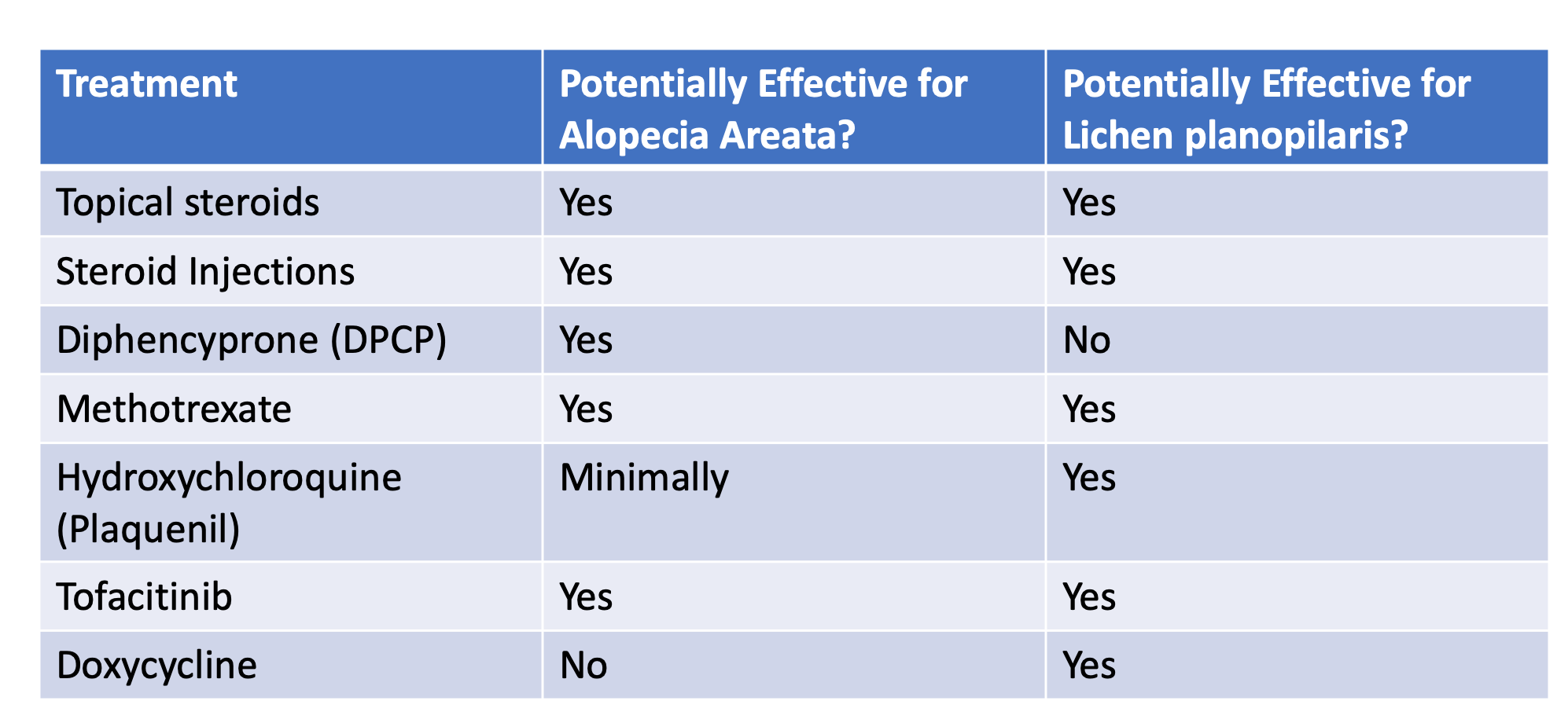
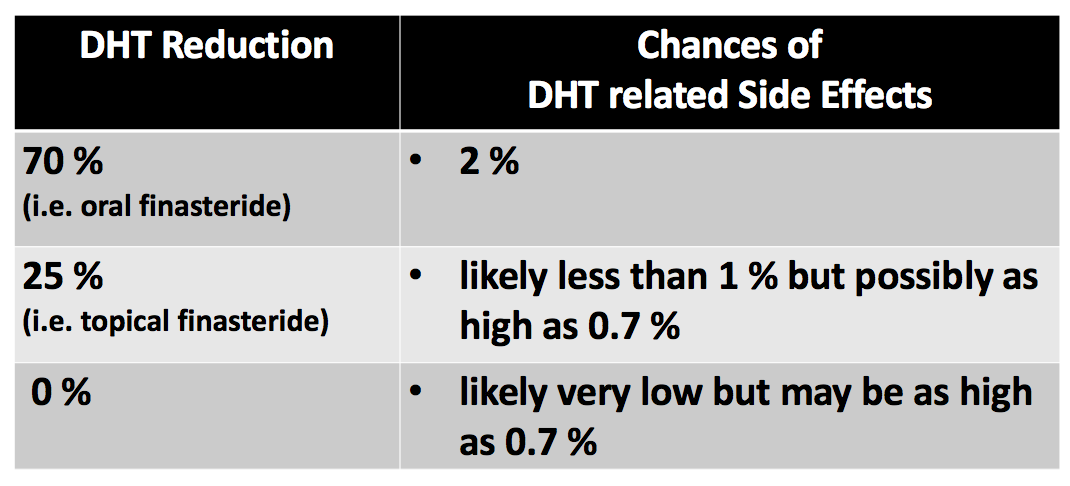
Higher Minoxidil Concentrations: Is More Always Better?
10 % Topical Minoxidil vs 5 % Topical Minoxidil: Which is better?
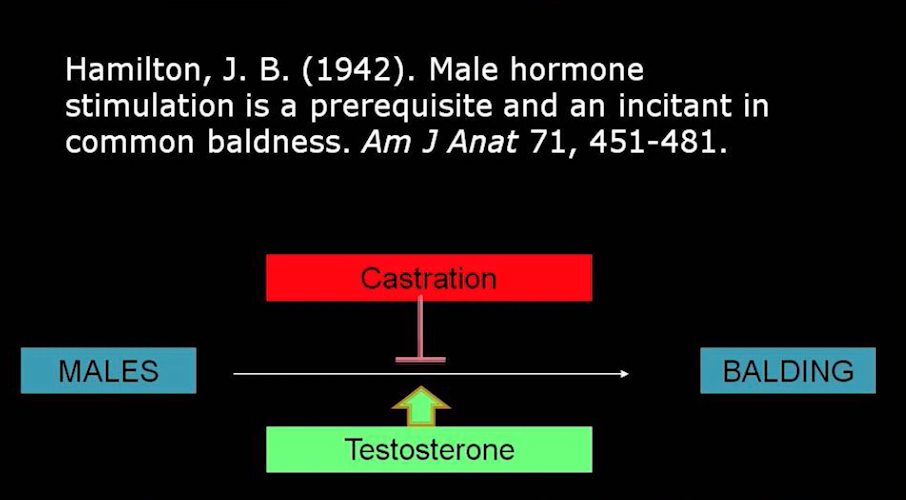
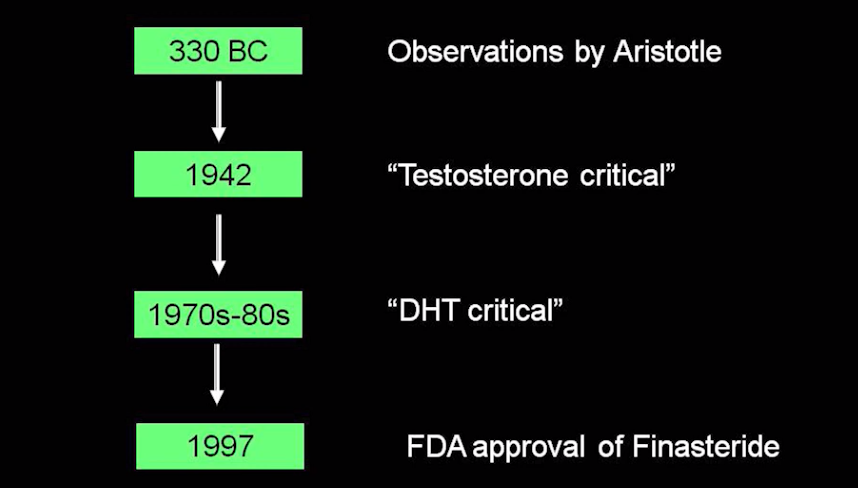
Minoxidil is FDA approved for treating androgenetic alopecia (male pattern balding and female pattern hair loss). It would seem logical to propose that if the drug minoxidil helps in the treatment of males and females with androgenetic alopecia that more minoxidil should help even more.
Researchers from Egypt set out to compare the efficacy and safety of 5% topical minoxidil with 10% topical minoxidil and placebo in 90 males with balding. The study was a double-blind placebo controlled randomized trial over 36 weeks. The study comprised three treatment groups: 1) study participants receiving 5 % minoxidil 2) study participants receiving 10 % minoxidil and 3) study participants receiving placebo.
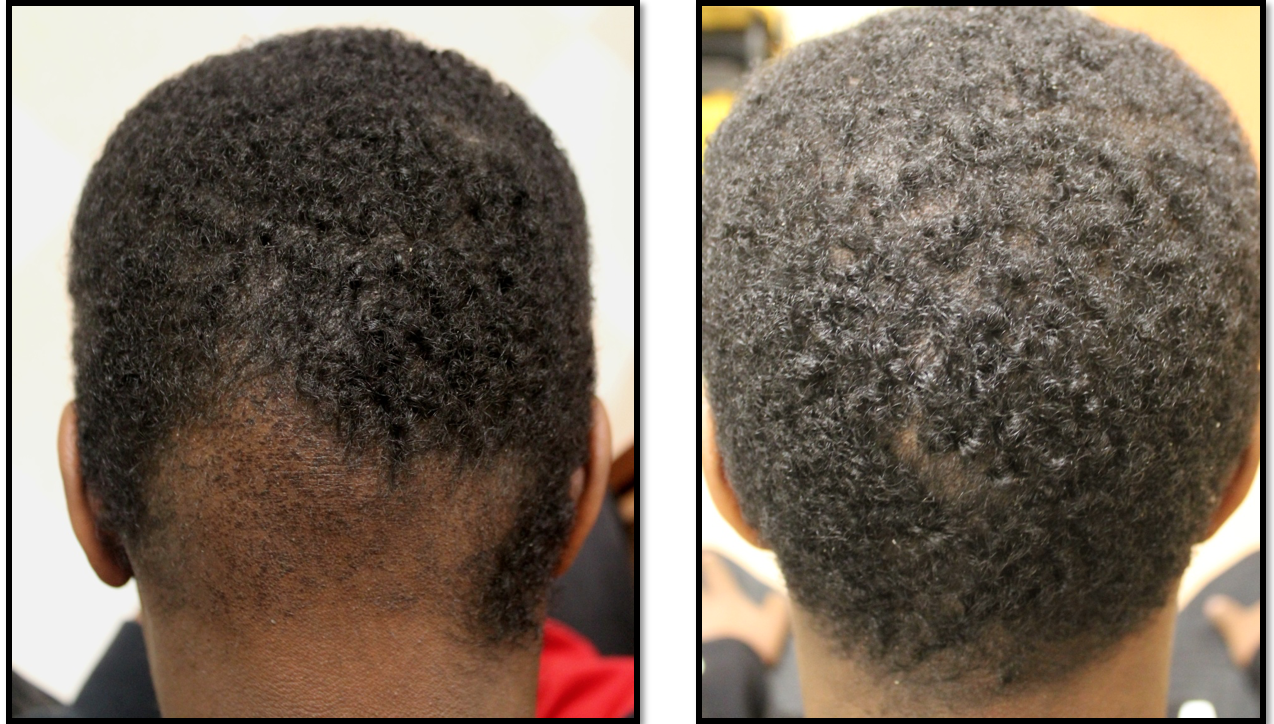
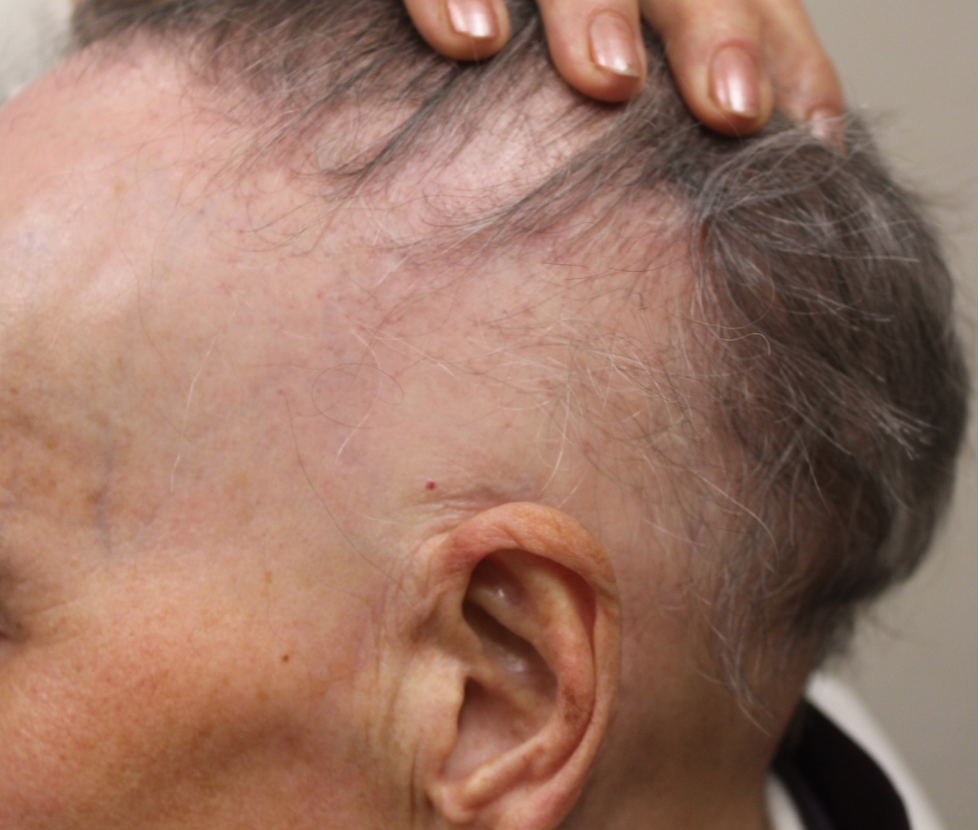
Surprisingly, after the 9 months, partipcants in the 5 % minoxidil group had higher vertex and frontal hair counts compared to study participants in the 10 % minoxidil group and the placebo group.
Conclusion
This was a nice study showing us that even after 40 years of studying minoxidil, we still have a lot to learn and a long way to go. Higher concentrations of minoxidil are not necessarily better - although more studies are clearly needed.
Reference
Ghonemy S et al. Efficacy and safety of a new 10% topical minoxidil versus 5% topicalminoxidil and placebo in the treatment of male androgenetic alopecia: a trichoscopic evaluation. J Dermatolog Treat. 2019 Oct 21:1-6. doi: 10.1080/09546634.2019.1654070. [Epub ahead of print]
This article was written by Dr. Jeff Donovan, a Canadian and US board certified dermatologist specializing exclusively in hair loss.